1Department of Hematology, Dhaka Medical College Hospital, Dhaka, Bangladesh; 2Massachusetts General Hospital, Boston, USA; 3Mugda Medical College, Dhaka, Bangladesh; 4Faridpur Medical College, Faridpur, Bangladesh
 10.3329/bmrcb.v45i2.42538
10.3329/bmrcb.v45i2.42538  0000-0003-2593-9703
0000-0003-2593-9703
Background: Bangladesh has started the journey of hemopoietic stem cell transplantation (HSCT) with successful autologous stem cell transplantation for multiple myeloma and lymphoma patients in Dhaka Medical College Hospital (DMCH). The purpose of this study was to describe the outcome of autologous stem cell transplanted patients for different haematological malignancies at DMCH under Department of Hematology and BMT unit.
Methods: Retrospectively analysed 30 post transplanted patients at this centre from March 2014 to December 2017. Data were obtained from a local software that was used for keeping all the patient related documents in BMT unit of DMCH.
Results: Two year Progression Free Survival (PFS) was 70.0% over a median observation period of 18 months. The cumulative incidence of relapse is 23.0% and transplant related mortality (TRM) in first 100 days was nil (0%). Demographic data were collected and complications of those patients.and challenges of stem cell transplantation in a developing country like Bangladesh were also identified.
Conclusion: Establishing stem cell transplant unit was a challenge in government hospital and it was successfully done at in Dhaka Medical College Hospital with the support of the government. Getting experience on autologous stem cell transplant, the next approach will be launching allogeneic stem cell transplant for haematological malignancies, aplastic anaemia and thalassemia patients.
Keywords: Hemopoietic stem cell transplantation (HSCT), Transplant related mortality (TRM), High dose therapy (HDT), Non relapse mortality (NRM)
Worldwide major advances in treatment for haematological malignancies have been observed in the past few decades. Based on the estimates of the worldwide incidence and mortality from all cancers GLOBOCAN reported that there were 6.5% haematological malignancies among 14.1 million new cases and 8.2 million deaths were from all cancers in 2012.1 Although Bangladesh has no population-based cancer prevalence registry, hospital based retrospective analysis reported that aacute myeloid leukaemia (AML), lymphoma and multiple myeloma are leading haematological malignancies of adult age group in our country.2 Haemopoietic stem cell transplantation is one of the curative option of treatment for thalassaemia, aplastic anaemia and many deadly haematological malignancies. Although the history of HSCT began in 1960s and it flared up in 1980s but Bangladesh started its journey of HSCT with the autologous stem cell transplant for multiple myeloma and relapsed or refractory lymphoma in Dhaka Medical College Hospital, the first and the pioneer HSCT centre in Bangladesh that was established in 2013 with the partnership collaboration between Bangladesh government and Massachusetts General Hospital (MGH), Boston, USA. The first autologous transplant was done for a multiple myeloma patient on 10th March 2014. The initial goal was to build a comprehensive program and centre of excellence for hematologic malignancies. The most important and crucial steps for establishing a stem cell transplant centre were (1) establishment and signing of bilateral memorandum of understanding (MOU) between DMCH and MGH, (2) appropriate infrastructure development, (3) human resource capacity building to make a core clinical team for transplant program and (4) financial considerations.3
The HSCT unit is approximately 7,000 square feet located at top floor of 9 storied new building DMCH-2 and includes 5 private inpatient rooms with HEPA filtration, separate apheresis area, irradiator for blood products, stem cell processing laboratory and general hematology labs, flow cytometry lab, separate pharmacy and shared inpatient rooms for receiving high dose chemotherapy. The Hematology and BMT department at DMCH consists of eight faculty members and twenty physician trainees, three specially trained technologists and 29 advanced trained nurses. The special nursing training was provided by ESNTP (Enhanced Specialized Nurse Training Program), a three-year bridging program between Government of Bangladesh and MGH, Boston USA, focused on the special nursing care regarding cancer patients and also care of bone marrow transplantation patients.4 As of December 2017, 30 ASCT have been done successfully in this centre without any transplant related mortality (TRM) in first 100 days.
It was retrospectively analysed 30 consecutive patients who underwent ASCT for the first time at bone marrow transplant Unit of DMCH from March 2014 to December 2017. The demographic data, response rate, progression free survival and mortality rate in first 100 days and beyond were evaluated. The data were obtained from a local software that is used to keep all patient related documents in BMT unit. Institutional review board certificate and ethical clearance were obtained as a part of institutional research protocol.
Of the total 30 patients multiple myeloma (MM)=11, relapsed/refractory non-hodgkin lymphoma (NHL)=10, relapsed/refractory Hodgkin lymphoma=06 and relapsed acute myeloid leukaemia (AML) =3 including one relapsed acute promyelocytic leukaemia (no matched donor available for AML).
| Characteristics | ||||
Age (median, range) |
36.56 (16-58) |
|||
M:F ratio |
26:4 |
|||
Disease |
Number (%) |
Salvage Rx/Mobilization |
Conditioning regimen |
|
MM |
11 (36.66%) |
VCD/RVD/+ Cy- GCSF |
High Dose Melphalan |
|
NHL |
DLBCL |
05 (16.66%) |
RICE/ + GCSF |
BEAM |
PTCL |
04 (13.33%) |
ICE/+ GCSF |
BEAM |
|
PBL |
01 (3.33%) |
DAEPOCH+ Bortezomib/+GCSF |
BEAM |
|
HL |
06 (20%) |
ICE (05)/GND (02)/ + GCSF |
BEAM |
|
AML |
03 (10%) |
HiDAC /+ GCSF |
Bu-Cy |
|
The M:F=26:4, average age was 36.56 with a range of 16-58 years for all cases and average age for MM patients was 49.54 years. All the myeloma patients were high risk in terms of ISS prognostic index, but cytogenetic profile was not of high risk panel for all patients. All the relapsed/ refractory lymphoma patients were of advanced stage (stage IIB or more) and variable IPI score (1-4).
Median observation period of survivors was 18 months (1.7-40 months) post transplant. Two year Progression Free Survival (PFS) was 70% over a median observation period of 18 months post transplant. Cumulative incidence of relapse was 23% and transplant related mortality (TRM)/NRM (Non Relapse Mortality) in first 100 days is nil (0%). Five MM patients died beyond 400 days on an average who had developed significant respiratory tract infection in addition to disease relapse beyond at least six months post transplant period. Five MM and two Hodgkin Lymphoma patients relapsed after stem cell transplantation and the earliest relapse was on five months post transplant. It could not be established any cytogenetic correlation in MM patients who relapsed as the sample number was too low to interpret the correlation between them.
All the myeloma patients were conditioned with high dose melphalan (200 mg/m2) and lymphoma patients were with BEAM standard regimen. Three relapsed AML was conditioned with standard Bu-Cy. No significant adverse reaction observed during chemotherapy administration. Major post transplant complications were bacteremia (8), pneumonia (3), Clostridium difficle colitis (2), CMV cystitis (1) and PRES (1) that were treated successfully. Overall Hospital stay was on average 15 Days. Average neutrophil and platelet engraftment was Day 10 and Day 11 respectively. All patients were supported with inj GCSF starting from D+3 post transplant until engraftment. Most patients neededone unit RCC (Red cell concentrate) and two units single donor platelet transfusion during their hospital stay on an average and all the blood products were irradiated before transfusion.
All the MM post transplant patients were being treated with 2 cycles of RVD followed by lenalidomide maintenance for 2 years.

Figure 1: Complications of 30 post transplant patients.
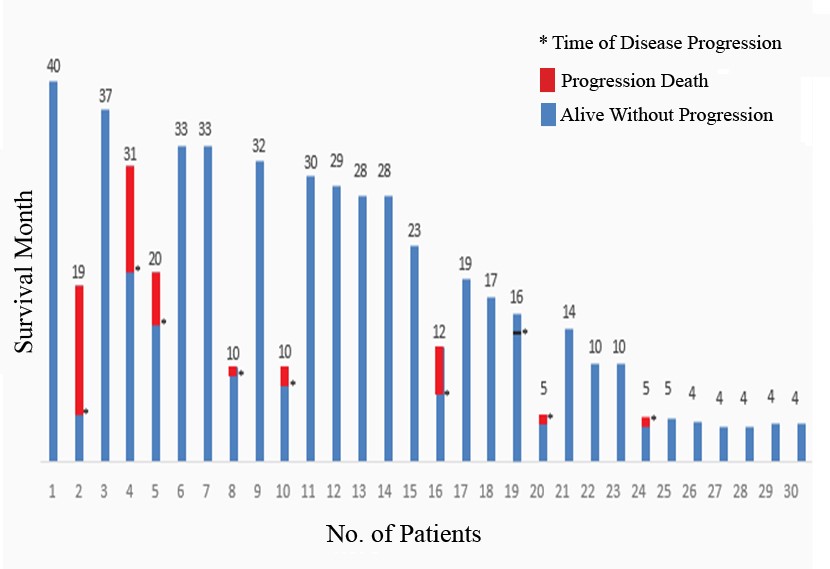
Figure 2: Survival Curve13
As a developing country it was a great challenge to start the stem cell transplantation first ever in Bangladesh emphasizing from the infrastructure development to the direct patient care at the biggest tertiary public Hospital, Dhaka Medical College Hospital (DMCH). Gratwohl et al conducted a global assessment of hematopoietic stem cell transplants and found that HSCT utilization rates were highly associated with countries with higher gross national income per capita, governmental health care expenditures, and human development index.5 With the continuous support of Government of Bangladesh it is now possible to run one of the most expensive and standard treatment in HSCT centre of DMCH with relatively low cost (around $7000-$10,000 ) comparing with the cost of that in developed countries and this is a great opportunity for the poor patients of haematological malignancy in the country.
Introduction of novel agents for multiple myeloma has led to the question of role of front line ASCT as a treatment of the disease. Considering the literature search during 1990s, before the era of novel agents, the several randomized trials have been found investigated the role of conventional chemotherapy versus high dose therapy (HDT) followed by ASCT in younger patients of MM and all of these data,the overall response rate was in favor of ASCT. Most of the trials showed significant benefit in favor of HDT followed by ASCT in terms of higher CR rate and higher rate of EFS. In the French IFM 90 trial autologous BMT versus conventional chemo the result showed higher CR rate (22% Vs 5%), better 5 year EFS (28% Vs 10%) and greater OS (52% Vs 12%).6 As a result the front line ASCT became the standard of treatment for young eligible MM patients since then.
In the last decade, introduction of novel agents thalidomide, lenalidomide and bortezomib have markedly improved the CR rate (VGRP 70 % in bortezomib containing regimen Vs 15 % in VAD) both before and after relapse MM without significant toxicities. It was stated that three drugs regimen incorporating bortezomib and dexamethasone with a third one with immune modulatory (Immid) is better than two drugs in MM although superiority of which three drugs combination is yet to be published (eg. RVD [lenalidomide, bortezomib and dexamethasone] and VCD [bortezomib, cyclophosphamide and, dexamethasone] both are of similar efficacy and potency).7-9
Cavo et al investigated in younger newly diagnosed MM patients to evaluate the efficacy of VTD (bortezomib, thalidomide, dexamethasone) Vs TD (thalidomide & dexamethasone) and the result showed clear benefit of VTD over TD in terms of better CR (38% Vs 23%), VGPR (79% Vs 58%) and PFS favors the VTD arm.10
In this centre majority (7 out of 11) of the myeloma patients were transplanted after first relapse or subsequent relapse, only 4 patients underwent upfront ASCT at 1st CR. It is to be mentioned that the median age of MM patients in Bangladesh is earlier than in western countriesand here it was also observed average age of myeloma patients is 49.54 years in this study.2 Thusthe ASCT can play an important mode of approach for majority of MM patients considering longer survival and better quality of life. Most of the relapsed/newly diagnosed patients were initially treated with thalidomide + dexamethasone or VAD (vincristine, adriamycin, dexamthasone) chemotherapy, although these protocols are not widely used in recent years due to availability of novel agents. All of them were in VGPR after reinduction with bortezomib containing three drug regimen, either VCD, VTD or RVD. All the myeloma patients were high risk in terms of International Staging System (ISS) prognostic index, but cytogenetic profile was not of high risk panel. As the sample size was too small it could not be concluded the co-relation of cytogenetics with ISS risk staging.
All the MM patients were collected the stem cells using cyclophosphamide and GCSF as soon as they were in at least VGPR.MM patients were conditioned with high dose Inj melphalan 200 mg/m2 and observed morbidity was less and the 100 days mortality was nil. Three patients of MM died beyond 400 days on average who had developed significant respiratory tract infection in addition to disease relapse around one year posttransplant period. It is also to be mentioned that relapse after autologous stem cell transplant can be managed with newer novel agents eg carfilzomib, pomalidomide, daratumumab etc., but due to unavailability and high expense of these medicine our patients could not be treated with those agents.
High dose chemotherapy followed by ASCT is the standard of care in chemo sensitive relapse or refractory lymphoma.11 In this new centre, it was treated 18 relapsed /refractory Lymphoma (NHL= 12, HD= 6) patients with BEAM conditioning followed by ASCT. Many relapsed/refractory lymphoma in the country are lately addressed to Hematologist and they are sometimes over treated with several agents that might have impact on chemo resistance. This factis reflected in lower number of transplanted patients at our centre. Patient’s chemo sensitivity was assessed after salvage chemotherapy (RICE, RDHAP, ICE or GND) that were being selected according to disease. Stem cell mobilization was preceded by inj GCSF following 2nd or 3rd salvage chemo protocol.11,12
The average CD34+ cell count collected for all 32 patients was 4.39 x106/kg (range 1.9-14 x106/kg) with mean viability rate of 96%. Engraftment, as defined by sustainedneutrophil count ≥ 0.5 x109/L and unsupported platelet count ≥20 x109/L occurred in all patients (range day 9-16).
Every patients at the centre gets ice chips during intravenous high dose Melphalan aiming to reduce the oral and upper GIT mucositis. The patients rarely had severe grade III-IV mucositis in the centre. We experience a little more CV line related blood stream infection in the neutropenic period but that did not hamper the engraftment as evidenced the average neutrophil engraftment was on Day +10 and platelet engraftment was on D+11. Eightout of 32 (25%) patients had positive blood CS for Pseudomonas (4), S. Epidermidis (2), Acenatobacter (1) and MRSA (1). Although CMV haemorrhagic cystitis is very rare in Autologous SCT, it was experienced one CMV haemorrhagic cystitis with CMV viremia in one patient who was treated with Inj Gancyclovir and was recovered well. Only one patient need ICU with ventilator support for severe pneumonia and she developed posterior reversible encephalopathy syndrome (PRES) and was managed successfully. Other expected toxicities nausea, emesis, loose stool, transient electrolyte imbalance were within tolerable limit among the patients and were managed accordingly.
Three relapsed AML (Acute Myeloid Leukaemia) was consolidated with standard dose busulfan-cyclophosphamide followed by ASCT as they had no matched donor and there is no scope of haploidentical transplant in Bangladesh. Till now two of them are in good remission status with very good quality of life.
On an average one unit RCC and 1 unit apheretic platelet were needed during the post transplant cytopenicperiod for all patients. All blood products were being irradiated and leucodepleted. All patients were given Inj GCSF routinely from D+3 until engraftment.
Conclusion
ASCT could significantly improve the overall survival and quality of life in MM and definitely a curative treatment for relapsed or refractory lymphoma. This small experience in the journey of transplant was relatively safe and without any transplant related mortality in first 100 post transplant days. Total 35 patients (five transplants were done after January 2018) have been transplanted with their autologous stem cells in this centre so far. The dedicated team work had enabled us to run the successful transplant program in government sector at DMCH. But the challenges are sustainable financial source and government support and development of human resource as a continuous process. As a new centrethe vision is to launch Allogeneic transplant very soon for aplastic anaemia, thalassemia and haematological malignancies and to establish this as a Centre of Excellence in near future.
References
- Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J Cancer. 2015 March 1; (5) 136.
- Hossain M, Iqbal M, Ahmed Khan M, Rabbani M, Khatun H, Munira S, et al. Diagnosed hematological malignancies in Bangladesh - A retrospective analysis of over 5000 cases from 10 specialized hospitals. Vol. 14. 2014.
- Yeh AC, Khan MA, Harlow J, Biswas AR, Akter M, Ferdous J, et al. Hematopoietic Stem-Cell Transplantation in the Resource-Limited Setting: Establishing the First Bone Marrow Transplantation Unit in Bangladesh. J Glob Oncol. 2016 Dec 21; (4):1–10.
- Barron A-M, Moran J, Nina SS, Harlow J, Gyawali M, Hossain F, et al. Building Specialized Nursing Practice Capacity in Bangladesh: An Educational Program to Prepare Nurses to Care for Oncology and Bone Marrow Transplant Patients in Dhaka, Bangladesh. J Glob Oncol. 2016 Dec 21; (4):1–6.
- Gratwohl A, Baldomero H, Aljurf M, Pasquini MC, Bouzas LF, Yoshimi A, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA. 2010 Apr 28; 303(16):1617–24.
- Attal M, Harousseau J-L, Stoppa A-M, Sotto J-J, Fuzibet J-G, Rossi J-F, et al. A Prospective, Randomized Trial of Autologous Bone Marrow Transplantation and Chemotherapy in Multiple Myeloma. N Engl J Med. 1996 Jul 11; 335(2):91–7.
- Cavo M, Pantani L, Pezzi A, Cavallo F, Petrucci MT, Raimondo FD, et al. Superior Efficacy of VTD over VCD As Induction Therapy for Autotransplantation-Eligible, Newly Diagnosed, Myeloma Patients. Blood. 2014 Dec 6; 124(21):197–197.
- Richardson PG, Weller E, Lonial S, Jakubowiak AJ, Jagannath S, Raje NS, et al. Lenalidomide, bortezomib, and dexamethasone combination therapy in patients with newly diagnosed multiple myeloma. Blood. 2010 Aug 5; 116(5):679–86.
- Kumar S, Flinn I, Richardson PG, Hari P, Callander N, Noga SJ, et al. Randomized, multicenter, phase 2 study (EVOLUTION) of combinations of bortezomib, dexamethasone, cyclophosphamide, and lenalidomide in previously untreated multiple myeloma. Blood. 2012 May 10;119(19):4375–82.
- Cavo M, Tacchetti P, Patriarca F, Petrucci MT, Pantani L, Galli M, et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stem-cell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet Lond Engl. 2010 Dec 18; 376(9758):2075–85.
- Philip T, Guglielmi C, Hagenbeek A, Somers R, Van der Lelie H, Bron D, et al. Autologous bone marrow transplantation as compared with salvage chemotherapy in relapses of chemotherapy-sensitive non-Hodgkin’s lymphoma. N Engl J Med. 1995 Dec 7;333(23):1540–5.
- Gisselbrecht C, Glass B, Mounier N, Singh Gill D, Linch DC, Trneny M, et al. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J Clin Oncol Off J Am Soc Clin Oncol. 2010 Sep 20; 28(27):4184–90.
- Akter M, Khan MA, Yeh AC, Spitzer TR, Ferdous J, Ara T, et al. The journey of stem cell transplantation in Bangladesh: a hike to the state of the art with collaboration between DMCH and MGH. Blood Adv. 2017 Dec 8;1(Suppl):62–4.